E-REB®–Rabeprazole Sodium INN
COMPOSITION
20 Tablet: Each enteric coated tablet contains Rabeprazole Sodium INN 20 mg.
PHARMACOLOGY
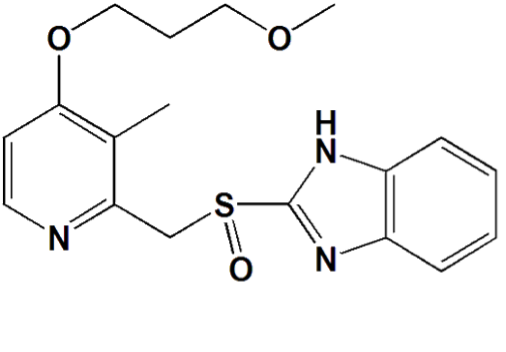
Rabeprazole Sodium (E-Reb) is an antiulcerant drug in the class of Proton Pump Inhibitors. Rabeprazole Sodium is a substituted benzimidazole which suppresses gastric acid secretion by inhibiting the gastric H+/K+-ATPase enzyme at the secretory surface of the gastric parietal cell. It blocks the final step of gastric acid secretion. It is an enteric coated tablet, because of its coated formulation, it is highly stable in the stomach and because of higher pKa value of Rabeprazole sodium, it provides faster onset of action. After oral administration of 20 mg, Rabeprazole is absorbed and can be detected in plasma by 1 hour. The effects of food on the absorption of rabeprazole have not been evaluated. Rabeprazole is 96.3% bound to human plasma proteins. It is primarily metabolized in the liver by Cytochrome P-450 3A (Sulphone metabolite) and 2C19 (Desmethyl Rabeprazole). Following a single 20 mg oral dose of Rabeprazole, approximately 90% of the drug is eliminated in the urine. The remainder of the dose is excreted in the feaces.
INDICATION
• Short-term treatment in the healing and symptomatic relief of duodenal ulcers and erosive or ulcerative Gastroesophageal Reflux Disease (GERD).
• Maintaining healing and reducing relapse rates of heartburn symptoms in patients with GERD.
• Treatment of day time and night time heartburn and other symptoms associated with GERD.
• Long-term treatment of pathological hypersecretory conditions, including Zollinger- Ellison Syndrome.
• Duodenal ulcer
• In combination with Amoxicillin and Clarithromycin to eradicate Helicobacter pylori.
DOSAGE & ADMINISTRATION
• Healing of Erosive or Ulcerative Gastroesophageal Reflux Disease (GERD): 20 mg to be taken once daily for 4 to 8 weeks. For those patients who have not healed after 8 weeks of treatment, an additional 8-week course may be considered.
• Maintenance of Healing of Erosive or Ulcerative Gastroesophageal Reflux Disease (GERD Maintenance): The recommended adult oral dose is 20 mg once daily.
• Treatment of Symptomatic Gastroesophageal Reflux Disease (GERD): The recom- mended adult oral dose is 20mg once daily for 4 weeks. If symptoms do not resolve completely after 4 weeks, an additional course of treatment may be considered.
• Healing of Duodenal Ulcers: The recommended adult oral dose is 20 mg once daily after the moming meal for a period up to 4 weeks. Most patients with duodenal ulcer heal within 4 weeks. A few patients may require additional therapy to achieve healing.
• Helicobacter pylori eradication to reduce the risk of duodenal ulcer recurrence:
Rabeprazole Sodium 20 mg Twice Daily for 7 Days
Amoxicillin 1000 mg Twice Daily for 7 Days
Clarithromycin 500 mg Twice Daily for 7 Days
• All three medications should be taken twice daily with the morning and evening meals. It is important that patients comply with the full 7-days regimen.
Treatment of pathological hypersecretory conditions including Zollinger-Ellison Syndrome: The dosage of Rabeprazole Sodium in patients with pathologic hypersecreto- ry conditions varies with the individual patient. The recommended adult oral starting dose is 60 mg once a day. Doses should be adjusted to individual patient needs and should continue for as long as clinically indicated. Some patients may require divided doses. Doses up to 100 mg QD and 60 mg BID have been administered. Some patients with Zollinger-Ellision syndrome have been treated continuously with Rabeprazole sodium for up to 1 year.
SIDE EFFECTS
Rabeprazole is well tolerable. But few side effects have been shown. More common-Headache, diarrhea, dry mouth, passing gas etc. Less common- Back pain, change in test & change in vision etc.
PRECAUTION
Proton pump inhibitors (PPIs) are possibly associated with increased incidence of Clostridium difficile-associated diarrhea (CDAD); consider diagnosis of CDAD for patients taking PPIs with diarrhea that does not improve. In liver disease may require dosage reduction. Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) reported with PPIs; avoid using for longer than medically indicated; discontinue if signs or symptoms consistent with CLE or SLE are observed and refer patient to specialist; most patients improve with discontinuation of PPI alone in 4-12 weeks; serological testing (e.g. ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations. Use of proton pump inhibitors may increase risk of salmonella and campylobacter infection. Reduce of symptoms does not eliminate presence of gastric malignancy; consider additional follow-up and diagnostic testing in adult patients who have suboptimal response or early symptomatic relapse after completing treatment with a PPI. Published observational studies suggest that PPI therapy may be associated with an increased risk of osteoporosis-related fractures of the hip, wrist, or spine; particularly with prolonged (greater than 1 year), high-dose therapy. Decreased gastric acidity increases serum chromogranin A (CgA) levels and may cause false positive diagnostic results for neuroendocrine tumors; temporarily discontinue PPIs before assessing CgA levels. Hypomagnesemia may occur with prolonged use (i.e., greater than 1 year); adverse effects, such as tetany, arrhythmias, or seizures, may result; in 25% of cases reviewed, magnesium supplementation alone did not improve low serum magnesium levels, and the PPI had to be discontinued; consider monitoring magnesium levels prior to initiation of PPI treatment and periodically. Monitor for increases in INR and prothombin time when coadministered with warfarin. Daily long-term use (e.g., longer than 3 years) may lead to malabsorption or a deficiency of cyanocobalamin. Acute interstitial nephritis reported in patients taking proton pump inhibitors. Concomitant use of proton pump inhibitors with methotrexate, primarily at high dose, may elevate and prolong serum concentrations of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities; in high-dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients.
CONTRAINDICATION
Rabeprazole Sodium is contraindicated in patients with known hypersensitivity to Rabeprazole or to any component in the product.
DRUG INTERACTION
Rabeprazole is metabolized by the Cytochrome P-450 (CYP-450) drug metabolizing enzyme system. Rabeprazole does not have clinically significant interactions with other drugs metabolized by the CYP-450 system, such as Warfarin and Theophylline given as single oral dose, Diazepam as a single intravenous dose, and Phenytoin given as a single intravenous dose. In normal subjects, co-administration of Rabeprazole 20 mg QD resulted in an approximately 30% decrease in the bioavailability of Ketoconazole and increase in the AUC and Cmax for digoxin of 90% and 29% respectively.
USE IN PREGNANCY & LACTATION
Rabeprazole is FDA pregnancy category OBÓ drug. No data is available on administration of Rabeprazole to pregnant women. However this drug should be used during pregnancy, only if clearly needed. There are no data on the excretion of Rabeprazole into the breast milk. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the benefit of the drug to the mother.
Use in pediatric patients: The safety and effectiveness of Rabeprazole in pediatric patients have not been established.
STORAGE
Store in a cool, dry place and away from light. Keep all medicines out of the reach of the children.
PACKAGING
E-Reb®: Tablet 20 mg: Each box contains 50’s tablets in alu-alu blister pack.